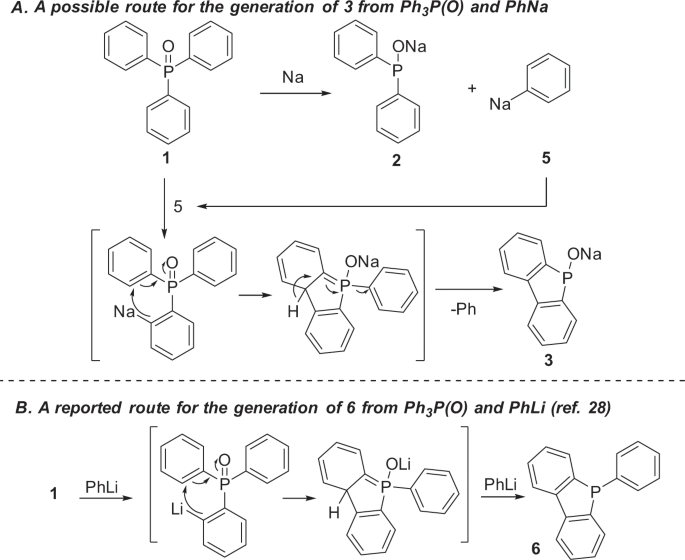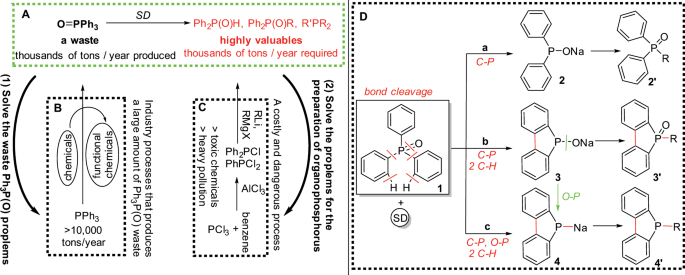
Conversion of triphenylphosphine oxide to organophosphorus via selective cleavage of C-P, O-P, and C-H bonds with sodium | Communications Chemistry

Strem, An Ascensus Company CAS# 14221-01-3. Tetrakis(triphenylphosphine) palladium(0), | Fisher Scientific

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

14221-01-3 Tetrakis (triphenylphosphine) Palladium - China Catalysts-Ligands; Catalystsfororganicsynthesis;, Classesofmetalcompounds; | Made-in-China.com

Photophysical Properties of Simple Palladium(0) Complexes Bearing Triphenylphosphine Derivatives | Inorganic Chemistry

The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -

Strem, An Ascensus Company CAS# 14221-01-3. Tetrakis(triphenylphosphine) palladium(0), | Fisher Scientific
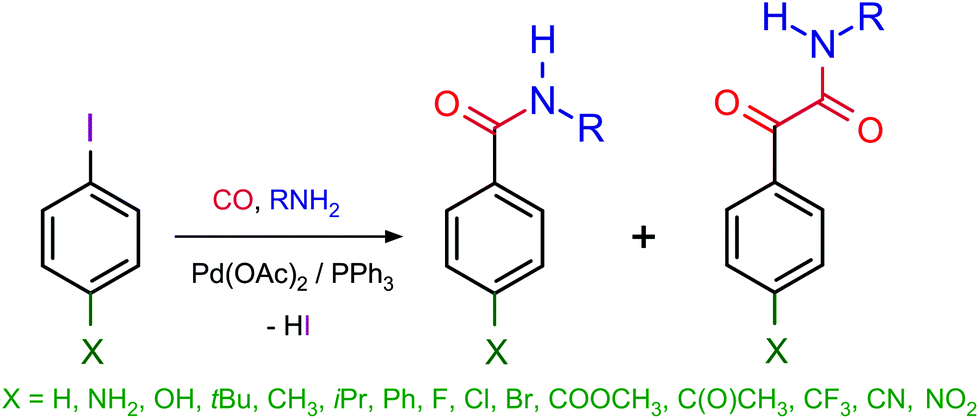
Viable pathways for the oxidative addition of iodobenzene to palladium(0)- triphenylphosphine-carbonyl complexes: a theoretical study - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT03642F

Hydrazine‐Free Facile Synthesis of Palladium‐Tetrakis(Triphenylphosphine) - Carrasco - 2019 - European Journal of Inorganic Chemistry - Wiley Online Library